Introduction
Materials and Methods
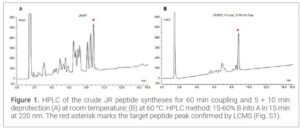
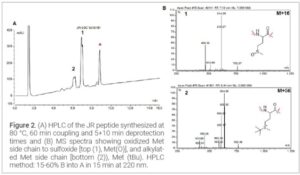
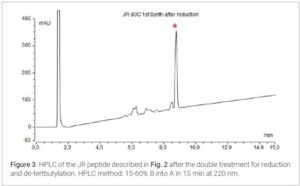
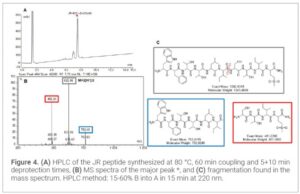
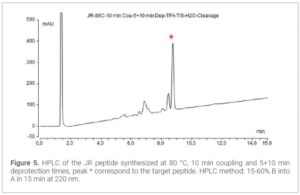
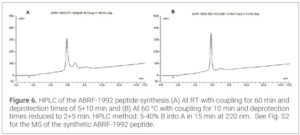
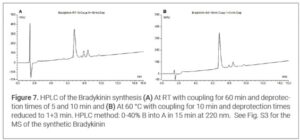
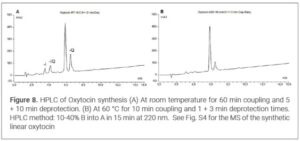
Results and Discussion
As a final remark, the heat exchanger allows for the close monitoring and management of synthesis temperature in ways that other heating technologies cannot. The obtention of higher crude purities enables the simplification of downstream purification and overall reduces the necessity for additional syntheses and materials, and more importantly, purifications to generate the desired mass of product.
- Muttenthaler M, King GF, Adams DJ, Alewood PF. Trends in peptide drug discovery. Nature reviews Drug discovery. 2021;20(4):309-25. https://www.nature.com/articles/s41573-020-00135-8
- Pintea A, Manea A, Pintea C, Vlad R-A, Bîrsan M, Antonoaea P, et al. Peptides: Emerging Candidates for the Prevention and Treatment of Skin Senescence: A Review. Biomolecules (Internet). 2025; 15(1). https://www.mdpi.com/2218-273X/15/1/88
- Zhang Y-M, Ye D-X, Liu Y, Zhang X-Y, Zhou Y-L, Zhang L, et al. Peptides, new tools for plant protection in eco-agriculture. Advanced Agrochem. 2023;2(1):58-78. https://www.sciencedirect.com/science/article/pii/S2773237123000035
- Das TN, Ramesh A, Ghosh A, Moyra S, Maji TK, Ghosh G. Peptide-based nanomaterials and their diverse applications. Nanoscale Horizons. 2025;10(2):279-313. https://pubs.rsc.org/en/content/articlelanding/2025/nh/d4nh00371c
- Merrifield RB. Solid Phase Peptide Synthesis. I. The Synthesis of a Tetrapeptide. Journal of the American Chemical Society. 1963;85(14):2149-54. https://pubs.acs.org/doi/10.1021/ja00897a025
- Ferrazzano L, Catani M, Cavazzini A, Martelli G, Corbisiero D, Cantelmi P, et al. Sustainability in peptide chemistry: current synthesis and purification technologies and future challenges. Green Chemistry. 2022;24(3):975-1020. https://pubs.rsc.org/en/content/articlehtml/2022/gc/d1gc04387k
- Martin V, Jadhav S, Egelund PHG, Liffert R, Johansson Castro H, Krüger T, et al. Harnessing polarity and viscosity to identify green binary solvent mixtures as viable alternatives to DMF in solid-phase peptide synthesis. Green Chemistry. 2021;23(9):3295-311. https://pubs.rsc.org/en/content/articlelanding/2021/gc/d1gc00603g
- Pacini L, Muthyala M, Aguiar L, Zitterbart R, Rovero P, Papini AM. Optimization of peptide synthesis time and sustainability using novel eco-friendly binary solvent systems with induction heating on an automated peptide synthesizer. Journal of Peptide Science. 2024;30(9):e3605. https://onlinelibrary.wiley.com/doi/full/10.1002/psc.3605
- Grunhaus D, Molina ER, Cohen R, Stein T, Friedler A, Hurevich M. Accelerated Multiphosphorylated Peptide Synthesis. Organic Process Research & Development. 2022;26(8):2492-7. https://pubs.acs.org/doi/10.1021/acs.oprd.2c00164
- Tamás B, Willi PL, Bürgisser H, Hartrampf N. A robust data analytical method to investigate sequence dependence in flow-based peptide synthesis. Reaction Chemistry & Engineering. 2024;9(4):825-32. https://pubs.rsc.org/en/content/articlelanding/2024/re/d3re00494e
- Pedersen SL, Tofteng AP, Malik L, Jensen KJ. Microwave heating in solid-phase peptide synthesis. Chemical Society Reviews. 2012;41(5):1826-44. https://pubs.rsc.org/en/content/articlelanding/2012/cs/c1cs15214a
- Alshanski I, Bentolila M, Gitlin-Domagalska A, Zamir D, Zorsky S, Joubran S, et al. Enhancing the Efficiency of the Solid Phase Peptide Synthesis (SPPS) Process by High Shear Mixing. Organic Process Research & Development. 2018;22(9):1318-22. https://doi.org/10.1021/acs.oprd.8b00225
- Sletten ET, Nuño M, Guthrie D, Seeberger PH. Real-time monitoring of solid-phase peptide synthesis using a variable bed flow reactor. Chemical Communications. 2019;55(97):14598-601. https://pubs.rsc.org/en/content/articlehtml/2019/cc/c9cc08421e
- Nandhini KP, Albericio F, de la Torre BG. 2-Methoxy-4-methylsulfinylbenzyl Alcohol as a Safety-Catch Linker for the Fmoc/tBu Solid-Phase Peptide Synthesis Strategy. The Journal of Organic Chemistry. 2022;87(15):9433-42. https://pubs.acs.org/doi/10.1021/acs.joc.2c01057
- Nandhini KP, Alhassan M, Veale CGL, Albericio F, de la Torre BG. Methionine-Containing Peptides: Avoiding Secondary Reactions in the Final Global Deprotection. ACS Omega. 2023;8(17):15631-7. https://pubs.acs.org/doi/10.1021/acsomega.3c01058
- Carpino LA, Krause E, Sferdean CD, Schümann M, Fabian H, Bienert M, et al. Synthesis of ‘difficult’ peptide sequences: application of a depsipeptide technique to the Jung–Redemann 10- and 26-mers and the amyloid peptide Aβ(1–42). Tetrahedron Letters. 2004;45(40):7519-23. https://www.sciencedirect.com/science/article/abs/pii/S0040403904017010?via%3Dihub
- Fields GB, Carr SA, Marshak DR, Smith AJ, Stults JT, Williams LC, et al. Evaluation of Peptide Synthesis As Practiced in 53 Different Laboratories. In: Angeletti RH, editor. Techniques in Protein Chemistry IV: Academic Press; 1993. p. 229-38. https://www.sciencedirect.com/science/article/abs/pii/B9780120587575500305?via%3Dihub


