Synthetic organic electrochemistry has gained significant attention from the chemical and pharmaceutical industries in recent years (1, 2). Electrolysis is seen as an enabling technology which can improve the efficiency and sustainability of chemical processes (3).
Electrochemical synthesis substitutes stoichiometric amounts of redox reagents by electrical current. Thus, hazardous oxidants like chromium(VI) salts, permanganates, or hypervalent iodine reagents can be avoided, reducing the handling of harmful chemicals and the generation of waste (4,5). Many electrochemical processes operate at ambient temperature and pressure, further improving the safety of manufacturing compared with conventional thermal reactions. An additional advantage of electrochemistry is that the lack of stoichiometric reagents also minimizes operations such as quenching and neutralization, which in turn reduces the use of water. The use of electrical current to drive chemical transformations can also significantly reduce the costs of manufacturing. Electrochemical energy is inexpensive compared to specialty chemicals such as oxidants or reductants. Additionally, eliminating the need for chemical reagents can also mitigate supply-chain volatility issues and minimize the expenses associated with transport and storage of hazardous materials. These features make electrochemical methods very attractive to pharmaceutical and fine-chemical manufacturers (6).
Implementation of synthetic electrochemistry in industrial settings is relatively straightforward when scale-up is carried out using flow electrolysis cells (7). The electrolysis efficiency of a cell depends on several key factors, including the ratio between the surface area of the electrodes and the volume of the reactor and the heat and mass transfer properties of the system (8). Flow electrolysis cells readily preserve these properties during scale-up, as small interelectrode gaps (often 1 mm or less) are maintained even for large scale preparations. Thus, electrochemical transformations developed on the lab scale can be translated to pilot and commercial scales with relative ease.
This manuscript provides an overview of the development of synthetic organic electrochemistry from the viewpoint of industrial manufacturing, from early successful implementation to recent trends and examples. Key considerations to make electrochemistry viable for manufacturing are also discussed.
Early Adoption in Industrial Organic Synthesis
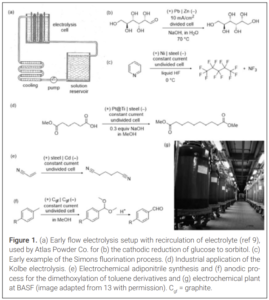
Pioneering work in the field of synthetic electrochemistry dates from the early and mid- 19th century, with the work of Faraday and the development of the classical Kolbe decarboxylative coupling. Not long after, several processes were implemented in industry. Notably, the use of flow electrolysis cells as the essential tool to scale up electro-organic transformations was already introduced nearly 100 years ago. An illustrative example is the flow process (Figure 1a) developed by the Atlas Powder Company in the 1930s for the manufacturing of sorbitol via cathodic reduction of glucose (Figure 1b)(9). The process featured a Zn cathode and a Pb-based anode, with an electrolyte consisting of an aqueous Na2SO4 solution. Several important electrochemical transformations involving organic compounds were developed from the 1950s to the 1970s. An example is the Simons fluorination process, the most common methodology to generate perfluorinated compounds. It uses HF as the fluorine source, which is attached to organic molecules under electrolytic conditions involving Ni electrodes. An early example disclosed by Simons described the multi-kilogram scale transformation of pyridine into perfluoropentane (Figure 1c), which generates NF3 as byproduct (10).
Notably, the classical Kolbe reaction was also translated to commercial scales. For example, a patent by Asahi Chemical Industry Co Ltd, published in the early 1970s, disclosed a process for the preparation of sebacic acid in which the key step was the electrochemical dimerization of the monomethyl ester of adipic acid (Figure 1d)(11). Two additional well-known electrochemical processes, also developed in the mid- 20th century, are the adiponitrile synthesis by Monsanto (Figure 1e)(12), and the anodic methoxylation of toluene derivatives by BASF (Figures 1f and 1g)(12). Both manufacturing processes are still used nowadays.
These early examples of industrial implementation of synthetic electrochemistry provided important foundations for modern electrochemical manufacturing. In particular, the advances in electrochemical engineering during the first half of the 20th and the development of flow electrolysis cells established the reactor technology principles used today.
Recent Contributions and New Trends – Increasing Complexity
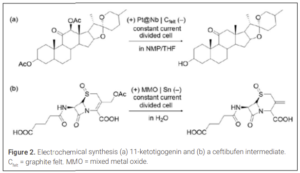
With the turn of the century, electrochemical processes involving more complex organic molecules were disclosed. Key to these advances were the development of important modern cell components from the 1960s to the 1980s, including mixed-metal oxide (MMO) electrodes and proton exchange membranes. For example, the Electrochemical Company described reductive deacetoxylation processes using flow electrolysis cells for the preparation of 11-ketotigogenin (Figure 2a)(14) and a key intermediate in the manufacturing of ceftibufen (Figure 2b)(15). Notably, the second example was carried out on a 2.8 kg scale using a repurposed ICI FM21-SP electrolyzer, originally designed for chlor-alkali production. The electrolyzer incorporated an MMO oxygen-evolving anode and a Nafion 350 cation-exchange membrane. Both reductive transformations showcased that conventional metal-based reductants can be successfully substituted by greener electrolysis methods.
Over the past 10 years, interest in synthetic organic electrochemistry in chemical manufacturers and the pharmaceutical industry has grown dramatically. A 2024 survey involving 15 major pharmaceutical companies (16) revealed that many are actively developing electrochemical methods for the synthesis of intermediates, with expectations of future scale-up. Notably, one of the main drivers behind this growing interest was not only the increasing demand for green and sustainable chemistry, but the development of disruptive synthetic routes, underscoring the perception of electrochemistry as an enabling technology. The survey also noted that a significant proportion of companies considered that there is a lack of adequate commercially available manufacturing equipment. Indeed, in many cases pilot and large-scale electrochemical manufacturing routes use either custom-made equipment or repurposed electrolyzers designed to operate under aqueous environments, which require the implementation of materials compatible with organic solvents. Simultaneously, electrochemical reactors with alternative designs to the traditional parallel plate electrolyzer, capable of processing solid-liquid mixtures owing to the incorporation of active mixers, have been recently described. These novel reactors, which can operate as a continuous stirrer tank electrochemical reactor (CSTER), are based on either static (17) or spinning electrodes (18).
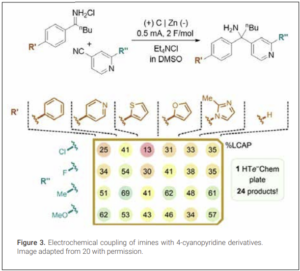
In medicinal chemistry, electrochemical methods are gaining traction as a tool for late-stage functionalization. Electrochemistry can provide excellent results for this purpose, particularly on small scale, as potentiostatic electrolysis can provide unique selectivity by applying the electrode potential matching the redox potential of the functional group of interest (19). Additionally, the development of several high throughout electrochemical experimentation platforms is enabling chemists to optimize reaction parameters and synthesize libraries of compounds efficiently (20). An example, disclosed by process chemists from Merck & Co., Inc., described the parallel synthesis of a library of 24 amines via the reductive coupling of imines with 4-cyanopyridine derivatives (Figure 3).
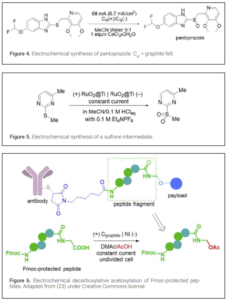
The resurgence of electrochemical methods in organic synthesis has promoted a significant amount of industry-academia collaboration in this research area, leading to numerous joint publications. Several of these articles deal with the development of key electrochemical steps in the synthesis of important active pharmaceutical ingredients. For instance, a recent contribution from Dr. Reddy’s laboratories in collaboration with academics reported an electrochemical method for the synthesis of pantoprazole from its thioether precursor (Figure 4)(21). The electrochemical procedure used inexpensive graphite electrodes and a simple electrolyte consisting of MeCN/water containing CaCl2, providing a green alternative to the conventional oxidants used to achieve this transformation. Notably, the anodic oxidation of a thioether has also been demonstrated on a kilogram scale by scientists from Merck (Figure 5)(22). The reaction was carried out in a mixture of MeCN and aqueous HCl. It used RuO2-based electrodes to selectively activate the chloride, which acted as a redox mediator. Kilogram scale experiments were carried out in a commercial flow electrolyzer.
Chemical manufacturers are also gradually adopting electrochemical synthesis as an alternative technology to conventional chemical manufacturing. In the CDMO sector, Lonza AG has published several articles describing the electrochemical synthesis of API intermediates, often in collaboration with academics. In a recent example, a green and scalable electrochemical procedure for the decarboxylative acetoxylation of Fmoc-protected peptides was described (23). This transformation is a key step in the manufacturing of linkers used in antibody-drug conjugates (ADCs). Notably, the electrochemical process avoided the use of stoichiometric amounts of toxic Pb(OAc)4. This is another example demonstrating the viability of electrochemical methods to prepare complex organic molecules. In this case, key to a successful process was the incorporation of a solvent with a relatively low oxidation potential, DMAc, which prevented degradation of the Fmoc protecting group.
Notably, contract manufacturers are also building manufacturing capabilities on commercial scale. As mentioned above, flow electrolysis cells are employed for this purpose, as flow electrochemical reactors can be readily scaled up. The German manufacturer Axplora has reported a current production volume of over 1000 tons of per year of 3 different active ingredients in 3 production sites in Germany and India using flow technology (Figure 7)(24).
Keys Factors for Industrial Implementation
The successful transition from conventional chemical methods to electrochemical approaches requires not only improvements in terms of safety, but also cost efficiency and reduction of waste generation. Electrochemical transformations from academic groups reported in the literature often use large amounts of supporting electrolytes exhibiting high molecular weights, such as tetraalkylammonium salts of tetrafluoroborate, hexafluorophosphate, or p-toluenesulfonate. The use of these salts, unless they can be recovered and recycled after the reaction, has a significant impact on the cost and the process mass intensity (PMI) of the electrochemical transformation (25). Additionally, many perchlorate salts used in laboratory electrosynthesis are potentially explosive, adding important safety concerns to otherwise inherently safe protocols. Thus, transition to low-molecular-weight inexpensive salts or acids, such as Na2SO4, CaCl2, NaCl, H2SO4, HCl, etc. is important. Apart from recycling strategies to reduce supporting electrolyte use, it has been shown that narrow gap flow electrolysis cells often allow the decrease of the supporting electrolyte concentration. Thus, the transfer of small-scale protocols to flow conditions on an early development stage, for reaction optimization purposes, is important to study this effect.
In addition to supporting electrolyte, the choice of solvent greatly affects the environmental footprint, safety, and cost of electrochemical processes. Many non-polar organic solvents, commonly used in synthesis, cannot be utilized in electrochemical processes due to their low dielectric constant, which provides poor conductivity even in the presence of a supporting electrolyte (26). Electrochemical reactions are often described using polar non-protic solvents such as dimethylformamide (DMF) or dichloromethane. However, the toxicity of these solvents makes them undesirable on large scale. Electrosynthesis in greener solvents, including water, ethanol or methanol, provides increased safety and sustainability. Recent research efforts have focused on identifying other green alternatives which maintain good electrochemical properties (high dielectric constant, broad electrochemical window) while improving sustainability. Bio-based solvents are being increasingly adopted in academic settings, opening the door to future adoption in industry. For example, it has been shown that dihydrolevoglucosenone (Cyrene®) has good electrochemical properties and can substitute DMF in electrolysis applications (27). The biomass-derived γ-valerolactone has also shown good electrochemical stability. It has been tested in organic electrolytes for capacitors and batteries (28) and could be employed as a solvent for electro-synthetic applications.
The cost and availability of electrode materials also have a significant influence on the industrial applicability of electrochemical synthesis. Noble metals such as platinum offer high stability and conductivity, as well as excellent electrochemical properties. However, their cost is prohibitive, limiting their use to small scale lab experimentation. Graphite and carbon felts are among the most used inexpensive electrode materials. They are chemically inert and possess a broad electrochemical window, making them suitable for a range of oxidative and reductive transformations. Stainless steel electrodes are commonly used as the cathode, also offering low cost, good conductivity, and mechanical stability. Alternatively, surface modification of inexpensive electrodes can also be employed. For example, platinized titanium has been used on commercial scale electrosynthesis. MMO-based electrodes are employed on very large-scale electrochemical processes, such as chloralkali production, and it has been demonstrated that they can also be utilized effectively for the electrochemical synthesis of organic compounds.
Conclusion
The renewed interest in synthetic organic electrochemistry over the past decade has the potential to transform the manufacturing of a wide range of chemicals and pharmaceuticals. By replacing stoichiometric redox reagents with electricity, electrochemical methods offer safer, cleaner, and often more cost-effective alternatives to conventional synthetic routes. These advantages align well with modern demands for greener synthetic routes, and the use of safer and less waste-generating technologies (30). Large scale electrolysis processes have been established for decades, incorporating the same principle of flow electrolysis technology used today. Flow reactors enable seamless scale-up from milligrams to gram-scales and to commercial manufacturing.
Despite the multiple advantages of electrochemical methods for synthesis, a broader adoption of electrochemistry as a synthetic tool still faces several challenges. These include the need for more accessible, robust and commercially available electrochemical equipment for large scale manufacturing. There is also a need for the implementation of cost-efficient supporting electrolytes, solvents, and electrode materials. Using inexpensive, recyclable salts and greener solvents is critical to improve the green chemistry metrics of promising electrochemical process developed in academia. Careful selection of these reaction components results in manufacturing processes which are superior to conventional protocols. Several examples of commercial electrochemical manufacturing of active ingredients have been disclosed in recent years, are many more are expected in the future.

References and notes
- Yan M, Kawamata Y, Baran PS. Synthetic Organic Electrochemical Methods since 2000: On the Verge of a Renaissance. Chem Rev. 2017;117:13230-319.
- Echeverria P-G, Delbrayelle D, Letort A, Nomertin F, Perez M, Petit L. The Spectacular Resurgence of Electrochemical Redox Reactions in Organic Synthesis. Aldrichimica Acta. 2018;51:3-19.
- Novaes LFT, Liu J, Shen Y, Lu L, Meinhardt JM, Lin S. Electrocatalysis as an enabling technology for organic synthesis. Chem Soc Rev. 2021;50:7941-8002.
- Frontana-Uribe BA, Little DR, Ibanez JG, Palma A, Vasquez-Medrano R. Organic electrosynthesis: a promising green methodology in organic chemistry. Green Chem. 2010;12:2099-119.
- Schaub T. Efficient Industrial Organic Synthesis and the Principles of Green Chemistry. Chem Eur J. 2021;27:1865-9.
- (a) Cohen B, Lehnherr D, Sezen-Edmonds M, Forstater JH, Frederick MO, Deng L, et al. Emerging reaction technologies in pharmaceutical development: Challenges and opportunities in electrochemistry, photochemistry, and biocatalysis. Chem Eng Res Des. 2023;192:622-37. (b) Laudadio G. The Role of Synthetic Organic Electrochemistry in the Technological Revolution of Pharmaceutical Industry. Chimia 2025;79:417.
- (a) Cantillo D. Recent advances in synthetic organic electrochemistry using flow systems. Curr Opin Electrochem. 2024;44:101459. (b) Tanbouza N, Ollevier T, Lam K. Bridging Lab and Industry with Flow Electrochemistry. iScience 2020;23:101720.
- Pletcher D, Green RA, Brown RCD. Flow Electrolysis Cells for the Synthetic Organic Chemistry Laboratory. Chem Rev. 2018;118:4573-91.
- Hales RA. Reducing sugars. US2289190A, July 7, 1942.
- Simons, JH. Production of Fluorocarbons: I. The Generalized Procedure and its Use with Nitrogen Compounds. J Electrochem Soc. 1949;95:47.
- Isoya T, Soya T, Kakuta R, Kawamura C. Process for the preparation of sebacic acid. US3896011A, July 22, 1975.
- Baizer MM, Campbell CR, Fariss RH, Rober J. Adiponitrile process. US3193480A, July 6, 1964.
- Hoormann D, Joerissen J, Puetter H. Elektrochemische Verfahren – Neuentwicklungen und Tendenzen. Chem Ing Tech. 2005;77:1363.
- Mazur DJ, Kendall PM, Murtiashaw CW, Dunn P, Pezzullo SL, Walinsky SW, Zung JB. Electrochemical Deacetoxylation: Synthesis of 11-Ketotigogenin. J Org Chem. 1996;61:405.
- Chai D, Genders D, Weinberg N, Zappi G, Bernasconi E, Lee J, Roletto J, Sogli L, Walker D, Martin CR, Menon V, Zelenay P, Zhang H. Ceftibuten: Development of a Commercial Process Based on Cephalosporin C. Part IV. Pilot-Plant Scale Electrochemical Reduction of 3-Acetoxymethyl-7(R)-glutaroylaminoceph-3-em-4-carboxylic Acid 1(S)-Oxide. Org Proc Res Dev 2002;6:178.
- Ferretti AC, Cohen B, Deng L, Diwan M, Frederick MO, Lehnherr D. Adoption of Electrochemistry within the Pharmaceutical Industry: Insights from an Industry-Wide Survey. Org Process Res Dev 2025;29:322.
- Griffin JD, Harper KC, Morales SV, Morrill WH, Thornton WI, Sutherland D, Greiner BA. A Scalable Solution to Constant-Potential Flow Electrochemistry. Org Process Res Dev 2024;28:1877.
- Petrović N, Malviya BK, Kappe CO, Cantillo D. Scaling-up Electroorganic Synthesis Using a Spinning Electrode Electrochemical Reactor in Batch and Flow Mode. Org Process Res Dev 2023;27:2072.
- Rafiee M, Mayer MN, Punchihewa BT, Mumau MR. Constant Potential and Constant Current Electrolysis: An Introduction and Comparison of Different Techniques for Organic Electrosynthesis. J Org Chem 2021;86:15866.
- Rein J, Lin S, Kalyani D, Lehnherr D. High-Throughput Experimentation for Electrochemistry. The Power of High-Throughput Experimentation: General Topics and Enabling Technologies for Synthesis and Catalysis (Volume 1). , 167-187
- Simon K, Bandichhor R, Beesu M, Manisha IS, Laudadio G, Kappe CO, Oruganti S, Budhdev RR, Cantillo D. Versatile Flow Electrochemical Methodology for the Manufacturing of Pharmaceutically Relevant Sulfoxides and Sulfones from Thioethers. ACS Electrochem 2025;1:1803.
- Bottecchia C, Lehnherr D, Lévesque F, Reibarkh M, Ji Y, Rodrigues VL, Wang H, Lam YH, Vickery TP, Armstrong BM, Mattern KA, Stone K, Wismer MK, Singh AN, Regalado EL, Maloney KM, Strotman NA. Kilo-Scale Electrochemical Oxidation of a Thioether to a Sulfone: A Workflow for Scaling up Electrosynthesis. Org Process Res Dev 2022;26:2423.
- Köpfler DM, Hanselmann P, Bersier M, Bovino C, Littich R, Wagschal S, Roberge DM, Kappe CO, Cantillo D. Solvent-Enabled Selective Electrochemical Decarboxylative Acetoxylation of Fmoc-Protected Peptides. ChemElectroChem 2025;12:e202500308.
- https://www.axplora.com/novasep-cdmo-an-axplora-company/electrochemistry (accessed on Oct 10, 2025)
- Cantillo D. Synthesis of Active Pharmaceutical Ingredients using Electrochemical Methods: Keys to Improve Sustainability. Chem Commun. 2022;58:619-28.
- Ramos-Villaseñor JM, Sartillo-Piscil F, Frontana-Uribe BA. Opportunities and challenges for water/organic solvents mixtures and renewable green solvents in organic electrosynthesis. Curr Opinion Electrochem 2024;45:101467.
- Ramos-Villaseñor JM, Sotelo-Gil J, Flores-Morales MP, Vasquez-Medrano R, Frontana-Uribe BA. Electrochemically relevant physical–chemical properties of tetraalkylammonium salts solutions in the renewable solvent dihydrolevoglucosenone (Cyrene®); electrochemical behaviour of some representative organic molecules. Green Chem 2025;(DOI: doi.org/10.1039/D5GC03356J).
- Teoh KS, Melchiorre M, Kreth FA, Bothe A, Köps L, Ruffo F, Balducci A. γ-Valerolactone as Sustainable and Low-Toxic Solvent for Electrical Double Layer Capacitors. ChemSusChem 2023;16: e202201845.
- Heard DM, Lennox AJJ. Electrode Materials in Modern Organic Electrochemistry. Angew Chem Int Ed 2020;59: 18866.
- Stephen HR, Röckl JL. The Future of Electro-organic Synthesis in Drug Discovery and Early Development. ACS Org Inorg Au 2024;4:571.


