Introduction
Every pharmaceutical contract development and manufacturing organization (CDMO) knows that quality is essential. But a CDMO should not merely comply with baseline current good manufacturing practices (cGMPs). Instead, it should embrace a holistic quality culture focused on excellence and continuous improvement.
For a CDMO to make this shift from mere compliance to quality excellence, it may be helpful to evaluate its systems through the lens of quality management maturity. The Quality Management Maturity (QMM) program initiated by the US Food and Drug Administration (FDA) in late 2023, provides an excellent framework for understanding what quality looks like at a mature organization, and what companies should consider as they strive to reach that maturity (1). The agency’s prototype assessment protocol suggests five practice areas: management commitment to quality; business continuity; an advanced pharmaceutical quality system; technical excellence; and employee engagement and empowerment (2). Below, we discuss the best practices and expectations for quality management maturity in these five areas.
Management commitment to quality
As the FDA’s protocol points out, management must set the tone and model a culture of quality. This starts with a quality policy that is communicated to all staff. The quality policy — described in ICH Q10 (3) — provides a road map that guides the entire organization—because the work required to ensure quality is not limited to the manufacturing and quality assurance teams; it requires commitment across every function and department.
To ensure that everyone understands the goals of the quality policy, quality metrics must be integrated into the performance goals of every employee, from upper management through warehouse staff, so that there is full alignment with the quality focus across the organization. Employees must also have the training and development required to meet and exceed these goals.
Recognition and awards for excellence are an excellent tool to keep staff motivated. Awards must be thoughtfully designed, to ensure that they reinforce the right actions. If awards are all focused on speed or efficiency, for example, staff will get the message that those criteria are more valued than ensuring quality.
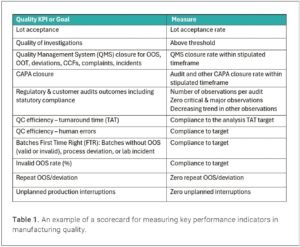
Performance in ensuring quality must also be measured at the organization level through a balanced scorecard. The scorecard should include factors such as the lot acceptance rate, the number of invalid out-of-specification (OOS) results, and the proper completion of all investigations (Table 1). Tracking performance against these key performance indicators (KPIs) fosters a culture of continuous improvement.
Management review of quality must also be effective. Per ICH Q10, management should have a formal process for reviewing the pharmaceutical quality system on a periodic basis (3). A sound approach is for quality data to be reviewed by multiple levels of management, such as weekly by the head of quality, monthly by the chief operations officer, and quarterly by the CEO. The results of those reviews should be used to initiate continual improvements.
Management must also provide the resources that are needed to meet quality goals and enable continuous improvement, such as new equipment and automation/digitization tools. The alignment of resources with quality objectives is a key indicator of quality management maturity.
Business continuity
The FDA’s QMM protocol also highlights the importance of business continuity in quality management. Supply chain security is the first element of a solid business continuity plan. A manufacturer must implement robust risk management to evaluate and mitigate potential supply disruptions. Redundancy of supply is essential, as is minimizing reliance on any one geography, to reduce the impact of political disruptions or natural disasters. Thorough vetting of suppliers, as well as building collaborative relationships with them, are also key. The European Medicines Agency’s Executive Steering Group on Shortages and Safety of Medicinal Products (MSSG) provides further recommendations in its guidance on strengthening the supply chains of critical medicines (4).
At the company’s own site, risk management and mitigation plans must address other possible disruptions. Wherever possible, a plan for emergency manual operation should be in place for automated systems. Every staff role must be backed up by at least one other person who is trained and prepared to do the work if needed. Quality control (QC) analysts should be qualified in multiple analytical techniques.
Facilities must also have strong data security, as well as continuity plans, for cyberattacks.
Regular and effective preventive maintenance plays a big role in avoiding unplanned production interruptions. Unplanned interruptions are an important KPI to track in the firm’s internal quality scorecard (see Table 1).
An Advanced Pharmaceutical Quality System
A robust manufacturing operation needs an advanced pharmaceutical quality system (PQS). Key elements of this practice area include quality risk management, the monitoring of process performance and product quality; change management; and leveraging corrective actions and preventive actions (CAPA).
Quality Risk Management
Ensuring pharmaceutical quality starts with quality risk management, which is described in detail in ICH Q9 (5). Through a thorough identification of potential risks to quality and the development of appropriate plans to avert or mitigate those threats, a manufacturing organization sets the stage for all other aspects of its quality system.
Monitoring Process Performance
An advanced PQS uses quality principles to ensure continued reliability in manufacturing operations, including continued process verification (CPV), which is the third stage of process validation, as defined in the US FDA’s process validation guidance framework (6). CPV is not an isolated activity; it builds on knowledge from earlier validation stages and reflects a lifecycle approach. It also recognizes that qualifying a process once is not enough; the manufacturer must consistently monitor the process to ensure it remains in a validated state.
Change Management
Change management is another critical aspect of an advanced PQS. Change management requires a clear standard operating procedure (SOP) that provides a structure for the process. As outlined in ICH Q10, major steps should include defining the proposed change, performing a thorough risk assessment of the change, and thoroughly following all stages of process validation (3).
Leveraging CAPA
Another important element is a robust CAPA system. Employing a digital platform for CAPA helps ensure accurate tracking and timely follow-up, as well as complete documentation. A company with quality management maturity leverages CAPA to improve its manufacturing operations.
The company should be learning from every out-of-specification result and every deviation investigation, identifying the true root cause, and then implementing measures to avoid the problem in the future. If an organization has multiple plants, the learnings from any CAPA process should immediately be applied to the other manufacturing units.
Companies can also learn from challenges faced by other pharmaceutical manufacturers, by subscribing to US FDA alerts for Form 483 citations. Such regulatory surveillance also highlights the evolving focus of FDA compliance teams, enabling the firm to stay up to date with the agency’s best practices.
Technical Excellence
Technical excellence is essential for quality management maturity. Technical excellence includes adopting advanced technologies that are fit-for-purpose. Companies should invest in upgrading manufacturing systems and technology to take advantage of automation, digitalization, and other technological advances. Examples of technologies that can help ensure quality are listed in Table 2.
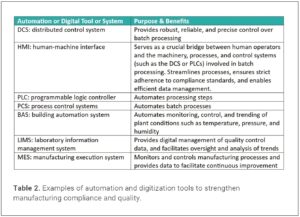
Digital management of quality control data, through a laboratory information management system (LIMS), eliminates the errors of manual data entry. This type of digital platform also ensures compliance with 21CFR11 regulations as well as the ALCOA+ principles for data integrity, which require that data be attributable, legible, contemporaneous, original, accurate, complete, consistent, enduring, and available to all who need them (7).
How well a manufacturing organization leverages data is another important measure of its quality management maturity. Digitization –through a LIMS or similar platform– facilitates integration of data across operations, enabling a holistic view of production, quality control, and related systems, such as supply chain management. This visibility supports better understanding of the operations and makes it easier to identify opportunities for process optimization.
Mature operations also support continuous improvement by seeking information externally, such as through conferences and training programs, including those offered by regulatory agencies. A CDMO can also learn from innovative customers, whose requirements can drive the adoption of new best practices.
Moving forward, forward-looking CDMOs, like other advanced pharmaceutical manufacturing organizations, will want to expand their use of automation and digitization to include even more advanced technologies and tools, such as model-based process development and digital twins, as well as GMP applications of artificial intelligence and machine learning. The report from the second Listen and Learn Focus Group meeting of the EMA’ Quality Innovation Group (QIG) provides examples of the application of such tools (8).
The EMA has also issued a reflection paper on the use of AI in the drug product lifecycle, including manufacturing (9). The US FDA’s draft guidance on the use of AI to support regulatory decision making also addresses examples related to GMP manufacturing, such as the use of AI for quality control of vial filling (10).
Employee Engagement and Empowerment
While automation and digitalization provide many advantages, advanced technologies alone, without engaged and empowered staff are insufficient for ensuring quality. Employee engagement begins with thorough training for new hires, on the employee’s specific duties, SOPs, and the reasoning and regulations behind the SOPs—showing the link between each person’s work and patient safety. Training must be followed by qualification through hands-on testing or operation, to ensure all procedures are fully understood.
In a mature organization, the quality culture ensures that employees are also comfortable speaking up about any concern related to quality or plant safety. Ongoing communication encourages this open culture. Regular one-on-one meetings between staff and their supervisors facilitate such communication, while skip-level meetings provide employees an opportunity to provide feedback they might not want to raise with their direct managers. A system for anonymous, confidential reporting provides a failsafe option.
Investment in employees’ continuous education, with regularly scheduled sessions, fosters a culture of continuous improvement and demonstrates the company’s commitment to excellence. A learning management system helps ensure that all staff actually participate actively in training and truly understand the information presented.
Achieving Quality Management Maturity: The Path to Excellence
Quality management maturity represents a strategic framework for CDMOs to evolve beyond baseline compliance toward organizational excellence. By thoroughly assessing and enhancing practices in management commitment, business continuity, pharmaceutical quality systems, technical capability, and employee engagement, CDMOs not only strengthen their compliance posture but also significantly improve performance, resilience, and reputation.
Ultimately, prioritizing quality maturity equips CDMOs to reliably deliver safe, high-quality pharmaceutical products, reinforcing trust with customers, regulators, and patients alike.
References and notes
- US Food and Drug Administration, CDER Quality Management Maturity (Silver Spring, Maryland). https://www.fda.gov/drugs/pharmaceutical-quality-resources/cder-quality-management-maturity (accessed 12 May 2025).
- US FDA, Center for Drug Evaluation and Research (CDER), CDER’s Quality Management Maturity (QMM) Program: Practice Areas and Prototype Assessment Protocol Development Maturity (Silver Spring, Maryland). https://www.fda.gov/media/171705/download?attachment (accessed 12 May 2025).
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), ICH Harmonised Tripartite Guideline, Pharmaceutical Quality System, Q10, Geneva, Switzerland (4 June 2008).
- European Medicines Agency (EMA), Executive Steering Group on Shortages and Safety of Medicinal Products (MSSG), MSSG recommendations to strengthen supply chains of critical medicinal products (Amsterdam, The Netherlands, 19 April 2024). https://www.ema.europa.eu/en/documents/other/mssg-recommendations-strengthen-supply-chains-critical-medicinal-products_en.pdf
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), ICH Harmonised Tripartite Guideline, Quality Risk Management, Q9R1, Geneva, Switzerland (18 November 2021).
- US FDA, Center for Drug Evaluation and Research (CDER), Guidance for Industry. Process Validation: General Principles and Practices (Silver Spring, Maryland, 2011).
- US FDA, Guidance for Industry, Data Integrity and Compliance with Drug CGMP Questions and Answers (Silver Spring, Maryland, 2018).
- EMA, Meeting Report. 2nd Listen and Learn Focus Group (LLFG) meeting, Quality Innovation Group (QIG) (Amsterdam, The Netherlands, 26 February 2024). https://www.ema.europa.eu/en/documents/report/meeting-report-second-listen-learn-focus-group-llfg-meeting-quality-innovation-group-qig_en.pdf
- EMA, Reflection paper on the use of Artificial Intelligence (AI) in the medicinal product lifecycle (Amsterdam, The Netherlands, 9 September 2024). https://www.ema.europa.eu/en/documents/scientific-guideline/reflection-paper-use-artificial-intelligence-ai-medicinal-product-lifecycle_en.pdf
- US FDA, Draft Guidance for Industry and Other Interested Parties. Considerations for the Use of Artificial Intelligence to Support Regulatory Decision-Making for Drug and Biological Products (Silver Spring, Maryland, USA, January 2025). https://www.fda.gov/media/184830/download
ABOUT THE AUTHOR
Ashutosh Kumar Sinha
Chief Quality Officer, Neuland Labs, Hyderabad, Telangana, India

Ashutosh Kumar Sinha serves as the Chief Quality Officer at Neuland Laboratories Ltd and brings more than 29 years of expertise in quality management systems. He has played a key role in designing and implementing comprehensive quality frameworks for various CDMO and generic pharmaceutical organizations. Mr. Sinha holds a postgraduate degree in pharmaceutical chemistry as well as an MBA in Total Quality Management from SMU.